By D. Kroeze MSc., CANNA Research
The primary colors
Researchers have traditionally distinguished seven colors. They are known as the colors of the rainbow, red, orange, yellow, green, blue, indigo and violet, often referred to as the primary and secondary colors. When put together, these primary colors create white light and only become visible when a ray of sunshine is split by a prism.
All objects can absorb the colors within the light, reflect them or allow them to pass through. Science tells us that an object usually adopts the color it isn’t able to absorb itself. Because plants cannot absorb the color green, they must themselves be green. There are those who claim that light measurements show that plants themselves emit light particles in the dark and actively help create their own color. Just like we do, plants sense the light and colors surrounding them, but in a different way!
Red light
Plants are sensitive to the color red in the light spectrum, a sensitivity that arises from the plant having what is called a red light photoreceptor. The receptor is a blue-green pigment called a phytochrome present in the cells of a plant. You could compare phytochrome with an eye that only senses red light.
Red light impacts a plant in many ways. Plants that are grown in plenty of red light are often large, but in general also tall with many branches. If the photoreceptor picks up a large quantity of natural red light, for example in summer when there’s plenty of natural red light, production of a plant hormone (metatopolin) is increased. This hormone prevents the chlorophyll in the plant from being broken down, so that it stays green in spring and summer; advantageously, this is exactly the time that the plant needs its chlorophyll to convert energy coming from the sun into sugar. Red light also influences a plant’s flowering and seed formation.

A prism refracts light into the primary colors
By comparing the quantity of one frequency of red light to the amount of another, far-red, present in the light, makes the plant decide whether to start flowering or not. The non-flowering period can be extended by exposing the plant to red-containing light during the dark period. This will, as a result, also extend the period of time before harvesting, which, of course, most growers would like to prevent. This also explains why it is a bad idea to enter the growing area when it’s dark for any time, even for a quick peek! The red color in light also influences flavor because it increases the concentration of special oils in plants.
Blue light
Plants see blue light as well as red light, using a photoreceptor that is called a cryptochrome. If there is plenty of blue light, as is the case in nature during autumn and winter, this receptor slows down the effect of a hormone called Auxin. This hormone is responsible for the plant’s stem and root growth. Auxin is also responsible for what is referred to as ‘apical dominance’, the phenomenon which causes the central stem to be dominant over side stems. A branch off a main stem would be dominant over its’ own side branches so it inhibits the development of axillary flowers. This causes the plant to create more side stems when exposed to bluish light and the plant stays shorter. This helps us to understand why plants exposed to bluish light are often short and bushy in appearance with a more robust structure.
Experiments with blue light resulted in plants that are wider than usual. This can be explained by the fact that more branches sprout because of diminished apical dominance.

Plants use the quantity of blue light to determine how far to open their stomas. The more blue light, the wider they open their stomas, resulting in an acceleration of their metabolism. High levels of blue light will increase metabolism, and as a consequence accelerate plant growth and development.
Blue light is also responsible for leaves growing towards the light. Blue light avoids the multiplication of leaves around the fruits. A shortage of blue light in the spectrum will quickly cause you to lose 20% of your harvest. Although opinions about this seem to differ, the optimum red to blue light ratio should be 5:1 in general.
Green light and the other colors
Plants are hardly sensitive to green light. As far as we know, they lack receptors for this color. This is probably the case because in practice plants do not absorb this color. Plants which are grown in green light only will be exceedingly weak and rarely grow old.
Clearly, plants only seem to sense those colors for which they have specific receptors. This is based on energy levels provided by each color. Plants are not blind but, up to a certain level, they are color blind. The way plants react to orange and yellow light is quite similar to the reaction on red light. This also accounts for indigo and violet which in reaction are similar to reactions on blue light.
‘Invisible’ light

The plant on the left is taller because it received less red light in relation to far-red light. The plant on the right was exposed to the usual red/far-red light relationship.
Although plants are a bit color blind, they do sense colors, more appropriately energy levels, that are entirely invisible to us humans. For example, plants can perceive far-red light. Plants often utilize the red to far-red relationship. A seed uses this relationship to determine whether to germinate or not. Plants also use that relationship to determine the number of other plants in the immediate surroundings.
Because plants absorb large amounts of red light whilst reflecting far-red light, there will be less red light present in a plant’s immediate surrounding if other plants are in the area too. Seeds will hold off germinating and the plants that are already in place will grow faster in order to emerge above the other plants to acquire sufficient light for their photosynthesis.
The fact that far-red light has the exact the opposite effect to that of red light makes it unsuitable as a light for growing. The traditional light bulb is a rich source of far-red light.
Ultra violet light (UV) also influences plants. Like is the case with blue light, plants perceive this color using the cryptochrome photoreceptor. It is unclear whether other photoreceptors can perceive UV light.
When increasing the quantity of UV light, the concentration of a purplish substance called Anthocyanin goes up. Anthocyanins protect plants against UV radiation, but also against micro-organisms trying to get in. The formation of Anthocyanin can often be seen where flaws occur such as a lack of oxygen. UV light not only damages the plant’s DNA and membranes, but immediately disrupts the process of photosynthesis. Therefore an excess of UV light is unhealthy for plants as well.

Left and right: far-red light is reflected by plants and can thus be used to determine the presence of other plants in the immediate vicinity. Plants can perceive far-red light.
Light is seeing
As we have seen, light is not just essential for plants when it comes to supplying energy for photosynthesis. Although this article only reveals a small part of everything there is to know about light, the colors in it and the effects they have on plants, we did see how plants use colors to regulate many of their processes. Plants are capable of perceiving those colors that matter to them. Those colors give the plant an indication of its general environment and its chances of survival and reproduction.
If your plants are to develop, grow and flower well, the composition of the light is at least as important as its quantity. Do not forget that a plant perceives the composition of both direct and indirect light. Indirect light here refers to the light that is reflected onto a plant by other objects such as walls or other plants.








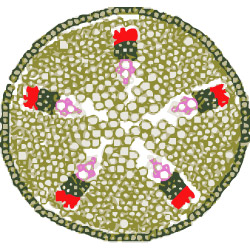 The phloem (red) is the living vascular tissue of the plant, through which mainly sugars and water are transported from the top down. Besides the phloem there is also xylem (pink), dead tissue that transports nutrients and water up from the roots.
The phloem (red) is the living vascular tissue of the plant, through which mainly sugars and water are transported from the top down. Besides the phloem there is also xylem (pink), dead tissue that transports nutrients and water up from the roots.
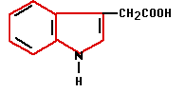 In the 1880’s Charles Darwin and his son Francis started experiments that finally confirmed the existence of plant hormones. They experimented with oats and the influence of light on the direction of growth. Auxin was the plant hormone whose processes were demonstrated during these experiments. Auxin is produced in the plant’s growing points both above the ground and in the roots. Auxin influences water absorption, cell division and cell stretching (it softens cell walls) among other things. Because auxin promotes the formation of roots on stems it is used in a variety of forms in rooting hormones.
In the 1880’s Charles Darwin and his son Francis started experiments that finally confirmed the existence of plant hormones. They experimented with oats and the influence of light on the direction of growth. Auxin was the plant hormone whose processes were demonstrated during these experiments. Auxin is produced in the plant’s growing points both above the ground and in the roots. Auxin influences water absorption, cell division and cell stretching (it softens cell walls) among other things. Because auxin promotes the formation of roots on stems it is used in a variety of forms in rooting hormones.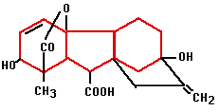 Gibberellin was first isolated in 1935 in Japan by Yabuta. The gibberellin was acquired from a fungus that had been the cause of reduced productivity for Japanese rice farmers for centuries. The gibberellin initially gave better growth but later in the season it caused sterile fruits. Generally speaking, gibberellins work as growth accelerators because of cell stretching and cell division. They ensure that seeds germinate and flowers form in plants that need long days. Gibberellin is often used in the cultivation of fruit to help unfertilized pears and apples develop fully.
Gibberellin was first isolated in 1935 in Japan by Yabuta. The gibberellin was acquired from a fungus that had been the cause of reduced productivity for Japanese rice farmers for centuries. The gibberellin initially gave better growth but later in the season it caused sterile fruits. Generally speaking, gibberellins work as growth accelerators because of cell stretching and cell division. They ensure that seeds germinate and flowers form in plants that need long days. Gibberellin is often used in the cultivation of fruit to help unfertilized pears and apples develop fully.
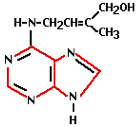 Cytokinin activity was first demonstrated in 1913. 30 years later it was discovered that a natural substance present in coconut milk was capable of helping plant cells multiply. Cytokinin was the responsible hormone for this. Cytokinin is known as the hormone responsible for cell division. It stimulates the metabolism and the formation of flowers on side shoots and as such is a counterpart to auxin. The cytokinin concentration is highest in young organs (e.g. seeds, fruits, young leaves and root tips). High concentrations in an organ or tissue will stimulate the transport of sugars to those tissues or organs.
Cytokinin activity was first demonstrated in 1913. 30 years later it was discovered that a natural substance present in coconut milk was capable of helping plant cells multiply. Cytokinin was the responsible hormone for this. Cytokinin is known as the hormone responsible for cell division. It stimulates the metabolism and the formation of flowers on side shoots and as such is a counterpart to auxin. The cytokinin concentration is highest in young organs (e.g. seeds, fruits, young leaves and root tips). High concentrations in an organ or tissue will stimulate the transport of sugars to those tissues or organs. The practical use of ethylene comes from the time of Old Egypt when figs were scored to make them ripen faster. In 1934 it was discovered that plants produce ethylene themselves, which enables them to regulate fruit ripening. Ethylene is the least complex plant hormone from the molecular point of view and is produced by all organs. It is a gaseous hormone which is transported via the spaces between plant cells. It is responsible for fruit ripening, inhibition of growth, and leaf abscission (shedding). Ethylene has a stimulating effect on flower formation with certain types of plants (i.e. pineapples, mangoes and lychees). Administering ethylene results in smaller plants and flowering finishes a lot quicker. The flowers ripen too quickly and consequently remain small.
The practical use of ethylene comes from the time of Old Egypt when figs were scored to make them ripen faster. In 1934 it was discovered that plants produce ethylene themselves, which enables them to regulate fruit ripening. Ethylene is the least complex plant hormone from the molecular point of view and is produced by all organs. It is a gaseous hormone which is transported via the spaces between plant cells. It is responsible for fruit ripening, inhibition of growth, and leaf abscission (shedding). Ethylene has a stimulating effect on flower formation with certain types of plants (i.e. pineapples, mangoes and lychees). Administering ethylene results in smaller plants and flowering finishes a lot quicker. The flowers ripen too quickly and consequently remain small.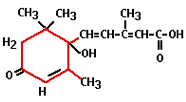 Abscisin was first isolated in 1963 and has the Latin word abscissio (breaking off) to thank for its name. This is because people thought that abscisin was responsible for the breaking off (shedding) of leaves and fruits, however, it was later shown that ethylene plays a much more direct role in this.
Abscisin was first isolated in 1963 and has the Latin word abscissio (breaking off) to thank for its name. This is because people thought that abscisin was responsible for the breaking off (shedding) of leaves and fruits, however, it was later shown that ethylene plays a much more direct role in this.